SOLUTION & TECHNOLOGY
OUR APPROACH
SYNSIGHT is a data-centric drug design company specialized in first-in-class small molecules, leveraging cell imaging and AI chemoinformatics to accelerate and secure drug discovery.
WORKFLOW
SYNSIGHT core team believes that the strength of AI in drug discovery comes from the quality and robustness of the DATA. SYNSIGHT Models are integrated models of disease, spanning multiscale experimental data and deep learning models.
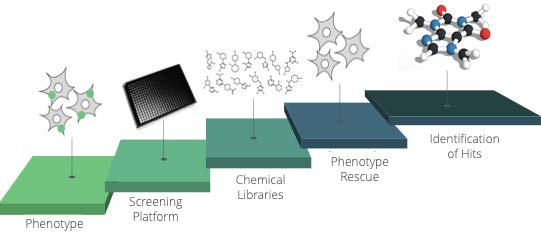
SYNSIGHT’s workflow is composed of iterative cycles mainly focused on back and forth between experimentation and chemoinformatics.
BIOLOGY
HIGH-CONTENT SCREENING
Phenotypic assays make it possible to evaluate the effects of molecules during screenings on a phenotype specific to a pathology. The data collected and analyzed are used to train the first generative models to propose new molecular entities satisfying a defined phenotype.
High-Content Screening allows to detect high-throughput interactions and to obtain massive and robust data by observing several thousand of cellular events. High-Content imager allows to acquire high-throughput and high-resolution fluorescence images (more than
60 000 images per day).

THE MICROTUBULE BENCH: The missing link between target and phenotype approach
SYNSIGHT developed the Microtubule Bench technology (MT bench®), making it evolves from a research tool to an industrialized cell testing to screen molecules by microscopy. Since 2021, this technology has made it possible to identify and quantify the modulations of small molecules on PPIs or between protein and nucleic acid.
The MT bench is a powerful technology to assess drug efficacy directly from inside the cell.
It can to provide qualitative and quantitative information within a cellular context. The MT bench is the unique cellular technology for targeting RNA with small molecules. It is the missing link to generate data and get a full view of the behavior of drug candidates.
This new bioassay represents the corner stone of SYNSIGHT platform and opens a wide range of challenging application such as PROTACs, downstream screening of kinases or aggregation into the cell.
ARTIFICIAL INTELLIGENCE & CHEMOINFORMATICS
Deep learning models rationally guide the generation of candidate molecules to access the most interesting molecules more quickly. These parametric optimization models are trained from a multi-scale data at the level of:
01. EFFICACY IN THE CELL (IMAGE ANALYSIS)
02. TARGET BINDING IN THE CELL (MICROTUBULE BENCH)
03. TARGET BINDING AT THE MOLECULE LEVEL (IN VITRO)
The impact of this method is important because, by using the cell phenotype as the only intermediate, it should offer a diversity of structures, which can induce the same phenotype.
It will make it possible to obtain molecules capable of producing the phenotype sought by new molecular pathways and unknown targets. It is proposed to develop GAN models that will reverse the phenotypic screening process, to predict candidate molecular structures from phenotypic images.
@SYNSIGHT2021. ALL RIGHTS RESERVED.

Genopole Entreprises
4 rue Pierre Fontaine
91058 EVRY
FRANCE




